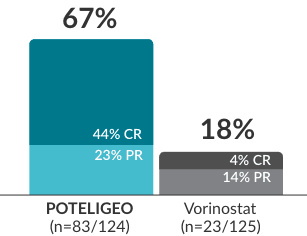
POTELIGEO achieved a higher response rate with a long duration of response (CR+PR) in blood1-3
Response rate in blood (confirmed response)a-c
(ad hoc analysis of secondary endpoint)
Duration of continued response (median)d
(post hoc analysis)

In skinb,e:
- POTELIGEO patients showed a 42% (n=78/186) response rate, compared to 15% in vorinostat patients (n=29/186) (ad hoc analysis of secondary endpoint)
- POTELIGEO patients (n=78) achieved a median 20.6-month duration of continued response, compared to 10.7 months in vorinostat patients (post hoc analysis)d
In lymph nodesf,g:
- POTELIGEO patients showed a 15% (n=21/136) response rate, compared to 3.7% in vorinostat patients (n=29/186) (ad hoc analysis of secondary endpoint)
- POTELIGEO patients (n=21) achieved a median 15-month duration of continued response; vorinostat patients were not estimable (post hoc analysis)d
In visceraf:
- No response was observed in viscera in either treatment group (POTELIGEO 0% [n=6]; vorinostat 0% [n=4])
- aResponse was defined by international response criteria for Mycosis Fungoides and Sézary Syndrome.4
- bResponses in blood and skin must have persisted for ≥4 weeks to be confirmed and were evaluated every 4 weeks during treatment.1,5
- cResponse in blood defined as >50% decrease in high blood tumor burden (B2), assessed by central flow cytometry.5
- d Duration of response was measured based on a post hoc analysis; a finding from the post hoc analysis cannot be used to demonstrate differences between treatments and may not be applicable to all patients initiating POTELIGEO.
- eResponse in skin defined as ≥50% clearance of skin disease without new tumors, evaluated using the modified Severity Weighted Assessment Tool (mSWAT).5
- fResponses in lymph nodes and viscera were evaluated at 4 weeks, then every 8 weeks for the first year, and every 16 weeks thereafter.2
- gResponses in lymph nodes defined as cumulative reduction ≥50% of measurable disease of each abnormal lymph node and no new abnormal lymph nodes, evaluated by computed tomography (CT) scans.5
- CR=complete response; PR=partial response
CTCL Expert Perspectives: POTELIGEO Efficacy and Safety
Hear Andrei Shustov, MD discuss the value of key endpoints used in the phase 3 MAVORIC trial, patient types appropriate for treatment with POTELIGEO, and the most common adverse reactions reported in MAVORIC.
POTELIGEO is administered in 28-day cycles1
Review dosing schedule- POTELIGEO [package insert]. Kyowa Kirin Inc., Princeton, NJ USA.
- Kim YH, Bagot M, Pinter-Brown L, et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): an international, open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2018;19(9):1192-1204.
- Data on file. Kyowa Kirin Inc., Princeton, NJ USA.
- Olsen EA, Whittaker S, Kim YH, et al. Clinical end points and response criteria in mycosis fungoides and Sézary syndrome: a consensus statement of the International Society for Cutaneous Lymphomas, the United States Cutaneous Lymphoma Consortium, and the Cutaneous Lymphoma Task Force of the European Organisation for Research and Treatment of Cancer. J Clin Oncol. 2011;29(18):2598-2607.
- Kim YH, Bagot M, Pinter-Brown L, et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): an international, open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2018;19(9):1192-1204. Supplementary appendix published online August 9, 2018. doi.org/10.1016/S1470-2045(18)30379-6
- Kim Y, Bagot M, Zinzani PL, et al. Safety of mogamulizumab in mycosis fungoides and Sézary syndrome: final results from the phase 3 MAVORIC study. Blood. 2019;134(suppl):5300[abstract].
- Cowan R, Scarisbrick JJ, Zinzani PL, et al. Efficacy and safety of mogamulizumab by patient baseline blood tumour burden: a post hoc analysis of the MAVORIC trial. J Eur Acad Dermatol Venereol. 2021;35(11):2225-2238.